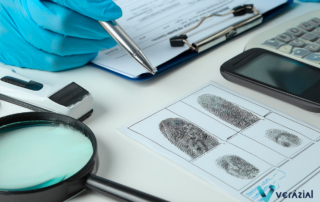
Use of biometric technology in criminal investigations
Accuracy in identifying suspects and analyzing evidence is crucial to avoid errors in criminal investigations. The implementation of biometric technologies, such as fingerprint, facial and iris recognition, allows for fast and reliable identification, improving the authenticity of evidence and strengthening trust in the judicial system.